|
Anti-structure
In crystallography, an anti-structure is obtained from a salt structure by exchanging anion and cation positions. For instance, calcium fluoride, CaF2, crystallizes in a cubic motif called the fluorite structure. The same crystal structure is found in numerous ionic compounds with formula AB2, such as ceria (CeO2), zirconia (cubic ZrO2), uranium dioxide (UO2). In the corresponding ''anti-structure'', called the antifluorite structure, anions and cations are swapped, such as beryllium carbide (Be2C) or lithium oxide (Li2O), potassium sulfate Potassium sulfate (US) or potassium sulphate (UK), also called sulphate of potash (SOP), arcanite, or archaically potash of sulfur, is the inorganic compound with formula K2SO4, a white water-soluble solid. It is commonly used in fertilizers, pro ... (K2SO4). Other anti-structures include: * anti- SnO2: Ti2N * anti- PbCl2: Co2P * anti- CdCl2: Co2N * anti- CdI2: Cs2O * anti- NbS2: Hf2S * anti- ReO3: Cu3N * anti- LaF3: Cu3P, Cu3As Referen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tin(IV) Oxide
Tin(IV) oxide, also known as stannic oxide, is the inorganic compound with the formula SnO2. The mineral form of SnO2 is called cassiterite, and this is the main ore of tin. With many other names, this oxide of tin is an important material in tin chemistry. It is a colourless, diamagnetic, amphoteric solid. Structure Tin(IV) oxide crystallises with the rutile structure. As such the tin atoms are six coordinate and the oxygen atoms three coordinate. SnO2 is usually regarded as an oxygen-deficient n-type semiconductor. Hydrous forms of SnO2 have been described as stannic acid. Such materials appear to be hydrated particles of SnO2 where the composition reflects the particle size. Preparation Tin(IV) oxide occurs naturally. Synthetic tin(IV) oxide is produced by burning tin metal in air. Annual production is in the range of 10 kilotons. SnO2 is reduced industrially to the metal with carbon in a reverberatory furnace at 1200–1300 °C. Amphoterism Although SnO2 is insolubl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
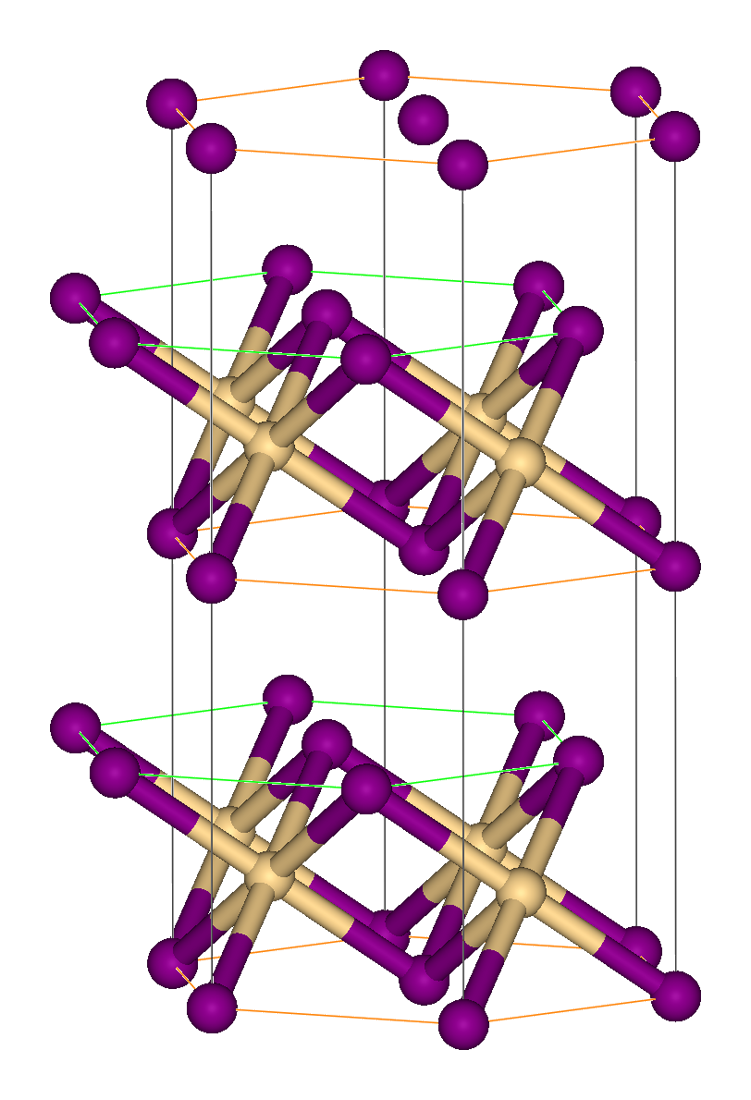
Lanthanum Fluoride
Lanthanum trifluoride is a refractory ionic compound of lanthanum and fluorine. The LaF3 structure Bonding is ionic with lanthanum highly coordinated. The cation sits at the center of a trigonal prism. Nine fluorine atoms are close: three at the bottom corners of the trigonal prism, three in the faces of the trigonal prism, and three at top corners of the trigonal prism. There are also two fluorides a little further away above and below the prism. The cation can be considered 9-coordinate or 11-coordinate. At 300 K, the structure allows the formation of Schottky defects with an activation energy of 0.07 eV, and free flow of fluoride ions with an activation energy of 0.45 eV, making the crystal unusually electrically conductive. The larger sized rare earth elements ( lanthanides), which are those with smaller atomic number, also form trifluorides with the LaF3 structure. Some actinides do as well. Applications This white salt is sometimes used as the "high-i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Rhenium Trioxide
Rhenium trioxide or rhenium(VI) oxide is an inorganic compound with the formula ReO3. It is a red solid with a metallic lustre that resembles copper in appearance. It is the only stable trioxide of the Group 7 elements ( Mn, Tc, Re). Preparation and structure Rhenium trioxide can be formed by reducing rhenium(VII) oxide with carbon monoxide at 200 °C or elemental rhenium at 400 °C. :Re2O7 + CO → 2 ReO3 + CO2 :3 Re2O7 + Re → 7 ReO3 Re2O7 can also be reduced with dioxane. Rhenium trioxide crystallizes with a primitive cubic unit cell, with a lattice parameter of 3.742 Å (374.2 pm). The structure of ReO3 is similar to that of perovskite (ABO3), without the large A cation at the centre of the unit cell. Each rhenium center is surrounded by an octahedron defined by six oxygen centers. These octahedra share corners to form the 3-dimensional structure. The coordination number of O is 2, because each oxygen atom has 2 neighbouring Re atoms., p. 1 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Niobium Disulfide
Niobium disulfide is the chemical compound A chemical compound is a chemical substance composed of many identical molecules (or molecular entities) containing atoms from more than one chemical element held together by chemical bonds. A molecule consisting of atoms of only one element ... with the formula NbS2. It is a black layered solid that can be exfoliated into ultrathin grayish sheets similar to other transition metal dichalcogenides. These layers exhibit superconductivity, where the transition temperature increases from ca. 2 to 6 K with the layer thickness increasing from 6 to 12 nm, and then saturates with thickness. References {{Sulfides Niobium(IV) compounds Sulfides Transition metal dichalcogenides Monolayers ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Caesium Oxide
Caesium oxide (IUPAC name), or cesium oxide, describes inorganic compounds composed of caesium and oxygen. Several binary (containing only Cs and O) oxides of caesium are known.. Caesium oxide may refer to: * Caesium suboxides (Cs7O, Cs4O, and Cs11O3) * Caesium monoxide (Cs2O, the most common oxide) * Caesium peroxide (Cs2O2) * Caesium sesquioxide (Cs2O3) * Caesium superoxide (CsO2) * Caesium ozonide Caesium ozonide (CsO3) is an oxygen-rich compound of caesium. It is an ozonide, meaning it contains the ozonide anion (O3−). It can be formed by reacting ozone with caesium superoxide: :CsO2 + O3 -> CsO3 + O2 The compound will react strongly w ... (CsO3) References {{Authority control Caesium compounds ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cadmium Iodide
Cadmium iodide is the inorganic compound with the formula CdI2. It is a white hygroscopic solid. It also can be obtained as a mono- and tetrahydrate. It has few applications. It is notable for its crystal structure, which is typical for compounds of the form MX2 with strong polarization effects. Preparation Cadmium iodide is prepared by the addition of cadmium metal, or its oxide, hydroxide or carbonate to hydroiodic acid. Also, the compound can be made by heating cadmium with iodine. Crystal structure In cadmium iodide the iodide anions form a hexagonal close packed arrangement while the cadmium cations fill all of the octahedral sites in alternate layers. The resultant structure consists of a layered lattice. This same basic structure is found in many other salts and minerals. Cadmium iodide is mostly ionically bonded but with partial covalent character. Cadmium iodide's crystal structure is the prototype on which the crystal structures many other compounds can be consid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cadmium Chloride
Cadmium chloride is a white crystalline compound of cadmium and chloride, with the formula CdCl2. This salt is a hygroscopic solid that is highly soluble in water and slightly soluble in alcohol. The crystal structure of cadmium chloride (described below), is a reference for describing other crystal structures. Also known are CdCl2•H2O and CdCl2•5H2O. Structure Cadmium chloride forms a layered structure consisting of octahedral Cd2+ centers linked with chloride ligands. Cadmium iodide, CdI2, has a similar structure, but the iodide ions are arranged in a HCP lattice, whereas in CdCl2 the chloride ions are arranged in a CCP lattice.N. N. Greenwood, A. Earnshaw, ''Chemistry of the Elements'', 2nd ed., Butterworth-Heinemann, Oxford, UK, 1997. Chemical properties Cadmium chloride dissolves well in water and other polar solvents. It is a mild Lewis acid. :CdCl2 + 2 Cl− → dCl4sup>2− Solutions of equimolar cadmium chloride and potassium chloride give potassium cadmium trichlo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lead(II) Chloride
Lead(II) chloride (PbCl2) is an inorganic compound which is a white solid under ambient conditions. It is poorly soluble in water. Lead(II) chloride is one of the most important lead-based reagents. It also occurs naturally in the form of the mineral cotunnite. Structure and properties In solid PbCl2, each lead ion is coordinated by nine chloride ions in a tricapped triangular prism formation — six lie at the vertices of a triangular prism and three lie beyond the centers of each rectangular prism face. The 9 chloride ions are not equidistant from the central lead atom, 7 lie at 280–309 pm and 2 at 370 pm. PbCl2 forms white orthorhombic needles. File:Cotunnite-3D-balls.png, Ball-and-stick model of part of the crystal structure of cotunnite File:Cotunnite-3D-ionic.png, Space-filling model File:Cotunnite-Pb-coordination-geometry-3D-balls.png, Coordination geometry of Pb2+ File:Cotunnite-Cl-coordination-geometry-3D-balls.png, Coordination geometry of Cl− File:Cot ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Potassium Sulfate
Potassium sulfate (US) or potassium sulphate (UK), also called sulphate of potash (SOP), arcanite, or archaically potash of sulfur, is the inorganic compound with formula K2SO4, a white water-soluble solid. It is commonly used in fertilizers, providing both potassium and sulfur. History Potassium sulfate (K2SO4) has been known since early in the 14th century. It was studied by Glauber, Boyle, and Otto Tachenius, Tachenius. In the 17th century, it was named ''arcanuni'' or ''sal duplicatum'', as it was a combination of an acid salt with an alkaline salt. It was also known as ''vitriolic tartar'' and ''Glaser's salt'' or ''sal polychrestum Glaseri'' after the pharmaceutical chemist Christopher Glaser who prepared it and used medicinally. Known as ''arcanum duplicatum'' ("double secret") or ''panacea duplicata'' in pre-modern medicine, it was prepared from the residue (''caput mortuum'') left over from the production of aqua fortis (nitric acid, HNO3) from nitre (potassium nitrate ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Crystallography
Crystallography is the experimental science of determining the arrangement of atoms in crystalline solids. Crystallography is a fundamental subject in the fields of materials science and solid-state physics (condensed matter physics). The word "crystallography" is derived from the Greek word κρύσταλλος (''krystallos'') "clear ice, rock-crystal", with its meaning extending to all solids with some degree of transparency, and γράφειν (''graphein'') "to write". In July 2012, the United Nations recognised the importance of the science of crystallography by proclaiming that 2014 would be the International Year of Crystallography. denote a direction vector (in real space). * Coordinates in ''angle brackets'' or ''chevrons'' such as <100> denote a ''family'' of directions which are related by symmetry operations. In the cubic crystal system for example, would mean 00 10 01/nowiki> or the negative of any of those directions. * Miller indices in ''parentheses'' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lithium Oxide
Lithium oxide ( O) or lithia is an inorganic chemical compound. It is a white solid. Although not specifically important, many materials are assessed on the basis of their Li2O content. For example, the Li2O content of the principal lithium mineral spodumene (LiAlSi2O6) is 8.03%. Production Lithium oxide is produced by thermal decomposition of lithium peroxide at 300–400 °C.Wietelmann, Ulrich and Bauer, Richard J. (2005) "Lithium and Lithium Compounds" in ''Ullmann's Encyclopedia of Industrial Chemistry'', Wiley-VCH: Weinheim. . Lithium oxide forms along with small amounts of lithium peroxide when lithium metal is burned in the air at and combines with oxygen at temperatures above 100 °C: :4Li + → 2. Pure can be produced by the thermal decomposition of lithium peroxide, , at 450 °C :2 → 2 + Structure Solid lithium oxide adopts an antifluorite structure with four-coordinated Li+ centers and eight-coordinated oxides. The ground state gas phase molecul ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |